Our news about the nutrition facts reform from the FDA has been spreading like wildfire! We dug deeper to find for you the timeline that nobody seems to be providing yet. After speaking at length with the FDA’s Deputy Director Siobhan DeLancey, here’s what to expect in the days (and weeks, and years…) to come regarding the new label update.

STEP ONE: 90-day Public Commentary (Opens today! See below for how to place your comment)
The label reform is now open to a 90-day public commentary period where the FDA is expecting to hear from a variety of groups and individuals from nutritionists, consumers, and food industry groups.
STEP TWO: Review of commentary (duration unknown)
The FDA must then review and consider those comments to evaluate any possible changes to the reform. They were unable to give us an exact timeline as it is dependent on the number and breadth of the commends received.
STEP THREE: Two-year implementation after final rule
After the FDA has issued a final ruling, they are proposing a two-year implementation period for products to comply with new industry standards. “But we expect many companies will put the new label on their product earlier than that, as we saw when the original nutrition facts label requirement came out,” says DeLancey.
The Cost
The FDA was unable to comment on their proposed budget, but word has it it will save us more than it will cost.
U.S. Nutrition Labels Revamp may save $20-30 bln in health-care costs over 20 yrs http://t.co/uTlVsnlJla via @EntMagazine by @Kate_H_Taylor
— TanyaBenedictoKlich (@TanyaKlich) February 27, 2026
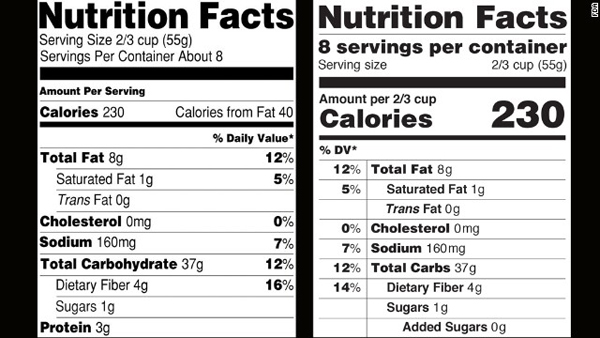
The New Label
To review, the new label will include updates that you can learn more about here. These include removal of calories from fat, new representation of serving size, and the inclusion of added sugars.
“We still have plenty of nutrition issues still on our plate.”
This was DeLancey’s response when asked why the delay in updating food labels. They note this is one of the first updates since food labeling began! Just think of how much has changed since then.
Steps in the Right Direction
While this new label won’t be a silver bullet to eliminate obesity, we are happy to finally see progress being made. “Our goal is to provide the public with the information they need and want, in an easily understood format, to help them make healthy choices,” DeLancey explains. It is still up to consumers to read the ingredients list, do their research, and take control of what they put in their bodies, but ‘for people looking to make a change in their diet,’ we want it to be helpful.”
Let your voice be heard! Comment on the new label reform, and the trans-fat ban, too!
The docket for label reform opens TODAY. Visit www.regulations.gov and enter this docket number in the keyword box: FDA-2026-N-1210
Remember the trans-fat ban that was open for 90-days of public commentary? Feedback is accepted until March 8th. Search docket # FDA-2026-N-1317.
To comment by mail*, send written comments to:
Division of Dockets Management
Food & Drug Administration, Room 1061
5630 Fishers Lane
Rockville, MD 20852
*Make sure the docket number is written at the top of each page of your comment!*
Also Read:
New Nutrition Label Gives Americans Reality Check
